About pH : pH standard solution
The pH measurement is a measurement of the relative value to the reference value, so the reference value must be determined precisely. This is the calibration. Since the electromotive force generated by the pH electrode varies depending on variations and the state of the surface of the glass film (such as stains), it must be corrected using a standard solution. Normally, ZERO point is calibrated with pH7 standard solution and SPAN is calibrated with another standard solution, and the pH meter stores the calibration curve.
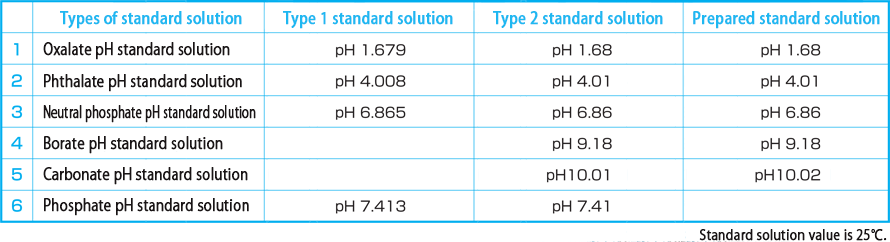
The standard solution is described in JIS. It is generally calibrated using a neutral phosphate pH standard solution of pH7(pH6.865), a phthalate pH standard solution of pH4(pH4.008), or a borate pH standard solution of pH9(pH9.180). JIS also describes oxalate pH standard solution (pH1.679), phosphate pH standard solution (pH7.413), carbonate pH standard solution (pH10.01) and saturated calcium hydroxide solution (pH12.45) as reference methods. These standard solutions are used according to the purpose.
Since the pH standard solution is a standard for pH measurement, Japan has established the Measurement Law Traceability System and JIS under the following conditions.
(1) Certified pH standard solution
A standard solution that has been tested by a public laboratory under the supervision of the government, and may only be type 1 (1/1000pH), type 2 (1/100pH), or type 2.
(2) Adjusted pH Standard Solution
Standards prepared by the methods specified in JIS Z 8802 are available in five types.
Class 1 pH standard solutions and Class 2 pH standard solutions are sold at designated sites certified by the government. Prepared pH standard solutions are prepared according to JIS and are commercially available without a national test. We sell only the prepared pH standard solution.