About dissolved oxygen (DO) : What is dissolved oxygen?
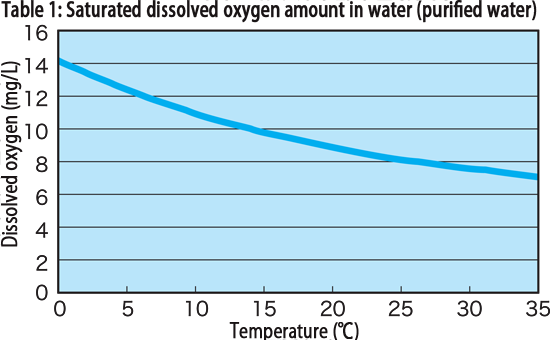
Dissolved oxygen (dissolved oxygen: DO) is the amount of oxygen dissolved in water (mg/L), and the amount dissolved in water increases as the water temperature decreases and the pressure increases. Under conditions of 1 atm and 25 °C, 8.26mg/L (saturated dissolved oxygen) of oxygen is thought to be dissolved. In addition, the oxygen concentration changes depending on the oxidation action of various coexisting substances and organic substances dissolved in water and the respiration action of microorganisms. The main source is oxygen in the atmosphere, but when algae in water grows, oxygen is released by photosynthesis and may temporarily become supersaturated. The relationship between the amount of saturated dissolved oxygen in water and water temperature is shown in Fig. 1.
Biochemical Oxygen Demand (Biochemical Oxygen Demand : BOD) is the amount of oxygen consumed when organic matter in water is decomposed by the action of micro-organisms and is an indicator of water pollution. If the organic fouling is large, the oxygen demand will be high, so the BOD will be a large number, and conversely, the clean water will have a lower BOD value.
