About oxidation-reduction potential : oxidation-reduction potential (ORP)
Oxidation refers to the process by which a substance loses electrons, and reduction refers to the process by which a substance obtains electrons, but this reaction occurs reversibly. When a platinum electrode and a reference electrode are placed, which are not affected by the solution in the redox system, a potential is generated between the surface of the electrode and the solution. This is called redox potential (Oxidation−Reduction Potential) (ORPs) and is represented by the following equation.
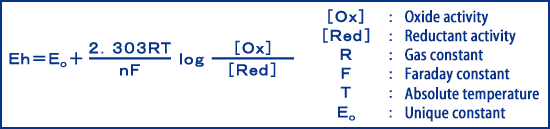
Eh is a value based on a hydrogen electrode as a standard in electrochemistry, but since the hydrogen electrode is complicated in configuration and impractical, ORP is measured with reference to the reference electrode and converted into a hydrogen electrode reference to obtain Eh.
As can be seen from the above equation, ORP is determined by the ratio of oxides to reductants, so if the ratio is constant, the same Eh is shown irrespective of concentration. In addition, when the oxide ratio is high, the potential fluctuates toward the positive side, and when the reduction ratio is high, the potential fluctuates toward the negative side.
