About oxidation-reduction potential : ORP measurement
1. ORP measurement
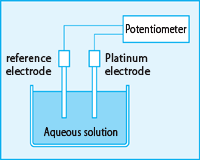
To measure ORP, use the mV range of the pH meter, or a potentiometer with an equivalent input resistance, a platinum electrode, and a reference electrode. Insert both electrodes into the aqueous solution to be measured as shown in the figure below, and read the potential difference obtained. In practice, a composite electrode is used in which a platinum electrode and a reference electrode are integrated.
Note that the reference electrode used in this case is different from the standard hydrogen electrode described above, and a silver/silver chloride electrode or a calomel electrode is used, so that the value obtained is not the correct value of Eh. To obtain the correct Eh value, add the value of the potential difference between the reference electrode and the standard hydrogen electrode used (see Table 1) to the measured value.
For example, if the measured value is 450mV when measuring an aqueous solution at 25°C using a saturated silver chloride comparator electrode,
Eh=450+199=649[mV]
ORP measurement is generally performed using platinum electrodes, but gold electrodes are used for liquids that are difficult to measure with platinum electrodes empirically, for example, for the management of treatment fluids for cyanogen wastewater and plating fluids.
2. Measurement accuracy
With a quinhydrone saturated solution, ORP measurement can be performed with an accuracy of ±10mV. However, the actual sample water is not as stable as the quinhydrone check solution. In particular, the thinner the oxidation-reduction substance concentration (the smaller the electrical conductivity), the more frequent the measurement is, and the more the electrode surface conditions change, the more likely measurement errors will occur. Therefore, the actual ORP measurement accuracy may be about ±50〜200mV.
3. Handling of platinum electrode
Platinum electrodes may change the surface state of the platinum electrode and affect the ORP measurement due to factors such as the oxidation of the surface to form an oxide film and the easy reaction with chlorine to form chloride. Therefore, it is necessary to regenerate the electrode surface accordingly.
4. Regeneration of platinum electrode
The surface of the platinum electrode changes due to the frequency of use and the measurement sample due to the formation of oxides and chlorides, and the correct measurement cannot be performed. The most effective way to restore performance is to polish the platinum electrode surface. (Use abrasives specified by us.)
