About pH : Glass Electrode and reference electrode
The glass electrode is equipped with a special glass film that responds to pH at the tip of the glass tube, and its interior consists of a constant concentration of internal liquid and an internal electrode that generates stable electromotive force. Since the glass film is a spherical thin film with a thickness of 0.1〜 0.3mm and φ10, it is easy to break and must be handled with care.
The pH measurement is not correctly measured unless only the electromotive force change in the glass film is detected. It is therefore necessary for the reference electrode to maintain a constant electromotive force and be stable in any liquid.
The silver-silver chloride internal electrode, a 3.3mol/L KCl solution, or an internal solution of a saturated KCl solution are selected and used under these conditions.
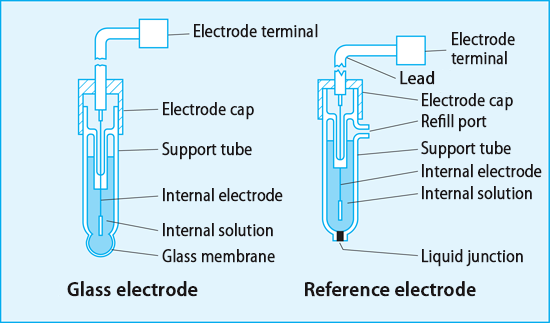
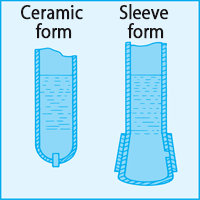
A liquid junction is provided at the tip of the reference electrode to maintain electrical connection with the liquid under test. Ideally, the liquid junction should be kept electrically connected and the internal fluid should flow out in small increments. Porous ceramic is generally used as the material of the liquid junction, but there is a sleeve-type liquid junction that uses glass rubbing as a liquid junction depending on the application.
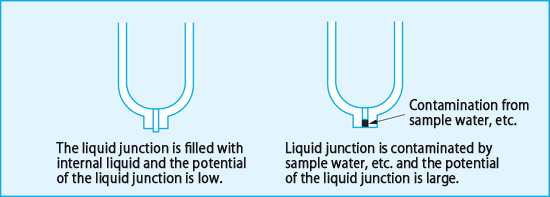
The liquid junction passes through a minute channel, and therefore, by the electrochemical principle it generates an inter-liquid potential due to the difference in mobility between anions and cations passing through it. Potassium chloride, which generates a very small amount of inter-liquid potential, is used as an internal solution for this purpose. When a liquid junction has contact with liquid under test, the liquid tends to gradually enter into the liquid junction due to the diffusion action. When the liquid to be measured accumulates in the liquid junction, an inter-liquid potential is generated, resulting in an error in measurement. In order to always fill the liquid junction with KCl solution, it is effective to increase the flow of the internal liquid. However, if the amount is too large, the internal liquid decreases quickly, and the KCl solution may enter the liquid to be measured and affect the pH concentration. Therefore, we have identified conditions for moderate flow and selected liquid junction materials.
The liquid-to-liquid potential differs depending on the liquid to be measured and is also affected by historical pollution of the liquid junction, etc., so it is difficult to theoretically specify it. Therefore, the potential generated in the liquid junction is collectively referred to as the liquid junction potential, including the theoretical liquid-to-liquid potential and the potential generated by contamination of the liquid junction.
For accurate pH measurement, the key point is to suppress the generation of liquid junction potential at a low level.